| Posted: Apr 08, 2009 | |
Nanotechnology catalysts offer prospect of commercially viable hydrogen fuel cells |
|
| (Nanowerk Spotlight) The high cost of today's leading fuel cell technologies, in particular the proton exchange membrane fuel cell, also known as polymer electrolyte membrane fuel cells (both resulting in the same acronym PEMFC), prevents their widespread use in cars and other applications. | |
| These fuel cells are powered by the electrochemical oxidation reaction of hydrogen and by the electroreduction of the oxygen (ORR) contained in air. They fuel cells run at relatively low temperature (<100°C) and therefore need catalysts to generate useful currents at high potential, especially at the electrode where oxygen is reduced (the cathode of the fuel cell). Presently, platinum-based electrocatalysts are the most widely used in PEM fuel cell prototypes. However, this metal is expensive due to its limited supply and its price is highly volatile. This creates one of the major barriers preventing commercialization of PEMFCs – the lack of suitable materials to make them affordable. | |
| Nanotechnology might provide solutions. In a previous Spotlight we introduced an example of how scientists are trying to develop nanoscale non-precious metal catalyst materials to improve the cost-performance ratio of PEMFCs (Nanotechnology is key to improving fuel cell performance). | |
| New work by this Canadian research team has now demonstrated that it is possible to significantly increase the catalytic site density of iron-based non-precious metal catalysts (NPMCs) to levels that were not thought possible before. The problem that this work resolves is that of the low activity of NPMCs compared to platinum-based catalysts. The best of these new NPMCs is more than 30 times more active compared to the previous best reported activity for NPMCs, and about 100 times more active than the majority of other NPMCs. Furthermore, their activity has reached about 1/10th the volumetric activity of state-of-the-art platinum-based catalysts (about 50 wt % platinum on carbon), which is the 2010 NPMC activity target set by the U.S. Department of Energy. | |
| While it has not been technologically possible to replace platinum-based catalysts with previous NPMCs in the past due to unacceptable loss of performance and efficiency of the system, this significant rise in activity now makes NPMCs a plausible alternative to platinum-based catalysts for PEMFCs. | |
| "The breakthrough in activity of these new NPMCs was realized via fundamental understanding of the processes involved in their synthesis acquired over the last 10 years," Jean-Pol Dodelet explains to Nanowerk. "Our new catalyst is based on inexpensive materials and simple processing steps that have resulted in activities that are 30-100 times higher than any previous NPMC reported in the literature. As a result, current density of a cathode made with our best iron-based electrocatalyst can equal that of a platinum-based cathode (0.4 mg platinum per square centimeter) at cell voltages ≥0.9V. However, below 0.9V, platinum-based cathodes still deliver higher current densities due to their better mass transport properties. " | |
| Dodelet is a professor at the INRS - Energy, Materials and Telecommunications at the Université du Québec in Canada. Together with his team – Michel Lefèvre, Eric Proietti and Frédéric Jaouen – he has managed to fabricate new NPMCs that differ from previous NPMCs in that they have increased catalytic activity resulting from an increase in active site density and/or in turn over frequency of each site. The team has reported their findings in the April 3, 2009 edition of Science ("Iron-Based Catalysts with Improved Oxygen Reduction Activity in Polymer Electrolyte Fuel Cells"). | |
 |
|
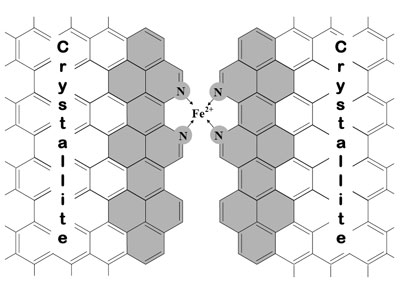
| Catalytic site in the micropores of the carbon support. Plan view of the presumed catalytic site and graphitic sheet growth (shaded aromatic cycles) between two crystallites after pyrolysis. (Image: Dr. Dodelet, INRS) | |
| Dodelet explains that the team's previous approach in the synthesis of NPMCs has been to use wet impregnation of carbon black with an iron precursor such as iron(II) acetate (FeAc), followed by a heat treatment in ammonia, the nitrogen precursor. During pyrolysis at temperatures of at least 800°C, ammonia partly gasifies the carbon support, resulting in a mass loss that depends on the duration of the heat treatment. The disordered domains of the carbon support are preferentially gasified. | |
| As a result, micropores are created in the carbon black particles. The mass loss at which maximum activity is reached corresponds to the largest microporous surface area for the etched carbon, which suggests that these micropores (width ≤2 nm) host most of the catalytic sites. The reaction of ammonia with the disordered carbon domains also produces the nitrogen-bearing functionalities needed to bind iron cations to the carbon support. | |
| "Thus, four factors have been identified as requirements for producing active iron-based catalysts for ORR: 1) disordered carbon content in the catalyst precursor, 2) iron, 3) surface nitrogen, and 4) micropores in the catalyst" says Dodelet. "By using this previous approach the catalytic activity may be improved by increasing the iron content. However, doing so increases the activity proportionally only up to approx. 0.2%, beyond which the activity levels off and eventually decreases. Surprisingly, the use of highly microporous carbon supports did not improve the activity relative to catalysts made with nonmicroporous carbon supports." | |
| Dodelet's team concluded that only the micropores created during heat treatment in ammonia host catalytic sites. The micropores in the off-the-shelf microporous carbon blacks do not bear the surface nitrogen necessary to form catalytic sites. Because these carbon blacks have little disordered carbon content, surface nitrogen is difficult to add during pyrolysis in ammonia. | |
| The INRS researchers' new approach capitalizes on the high micropore content of microporous carbon blacks and overcomes the limitations resulting from their lack of disordered carbon by filling their micropores with a mixture of pore filler and iron precursor. Doing so creates a catalyst precursor that complies with the four factors required for producing active NPMCs, as described above. To overcome the limitation of solubility and/or adsorbability associated with the impregnation method, they used planetary ball-milling to fill the pores of the microporous carbon support with pore fillers and iron precursor. Planetary ball-milling uses both friction and impact effects to force the filler materials into the pores of the carbon support while leaving its microstructure relatively unaffected. | |
| This work has advanced the field of PEMFC electrocatalysis by overcoming the first of the three key obstacles faced by NPMCs for ORR in PEMFCs: their low activity compared to platinum-based catalysts. The second is their poor stability and the third is the limited current density of NPMC electrodes – which are necessarily thicker than platinum-based electrodes – due to mass transport losses. | |
| "Before we developed these new NPMCs, their greatest shortcoming was arguably their low activity" says Dodelet. "The activity breakthrough realized by our new NPMCs addresses this problem. While there is still room for further activity improvement it is no longer the main challenge because NPMC-based electrodes are now capable of equaling the kinetic current density of platinum-based electrodes. Instead, the focus must now shift to improving their stability and optimizing the electrodes to minimize mass transport losses and increase power densities." | |
| Today, most NPMC catalysts are unstable. For automotive fuel cells, stable catalysts for at least 5000 hours of operation are needed. Some NPMCs have shown stability for 100 hours ("A class of non-precious metal composite catalysts for fuel cells"), but their activity is lower and the reason for their stability is still poorly understood. | |
| For the new NPMCs developed by Dodelet and his team, resolving the stability problem has now become the main challenge. The solution to this problem requires a fundamental understanding of the degradation mechanism for these catalytic sites. Dodelet is confident that stability can be resolved independently, just as activity was. | |
| In addition to resolving the stability challenge, electrodes made with these new NPMCs – which are necessarily thicker (50-100 µm) than platinum-based electrodes (10-15 µm based on a metal loading of 0.4 mg platinum per square centimeter) due to their lower activity – must be optimized for mass transport to allow for higher power densities. Again, the scientists believe that this problem may be resolved by better electrode fabrication techniques and materials. | |
| Although detailed cost analysis of these new iron-based electrocatalysts was not performed, their principal elements are carbon, nitrogen, and iron and they don't require expensive precursors or processing steps. Their manufacturing cost is conservatively estimated to be at least two orders of magnitude lower than that of current platinum-based ORR catalysts for PEMFCs. Says Dodelet: "If the remaining obstacles with these NPMCs can be overcome, our new catalysts could significantly reduce the overall cost of PEMFCs." | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
