| Posted: Sep 03, 2008 | |
My corona! Novel spectroscopic technique could revolutionize chemical analysis |
|
| (Nanowerk Spotlight) Laser-based analytical techniques such as Raman spectroscopy, fluorescence spectroscopy or the state-of-the-art laser-induced breakdown spectroscopy (LIBS) are highly sophisticated techniques to analyze minute amounts of matter with regard to its structure, elemental composition, and other chemical properties. LIBS has been shown to be capable of analyzing extremely small samples with high sensitivity – nanoliter volumes with levels of detection in water of part per million. LIBS works by focusing short laser pulses onto the surface of a sample to create a hot plasma with temperatures of 10,000 - 20,000 °C. The plasma emits radiation that allows the observation of the characteristic atomic emission lines of the elements. On the downside, LIBS is complicated by the need for multiple laser pulses to generate a sufficiently hot plasma and the need for focusing and switching a powerful laser, requiring relatively large and expensive instruments. | |
| New research coming out of Drexel University has now shown that light emitted from a new form of cold plasma in liquid – field emission generated, highly non-equilibrium and high energy density – permits Optical Emission Spectroscopy (OES) analysis of the elemental composition of solutions within nanoseconds from femtoliter volumes. | |
| "We were able to generate, for the first time, a nonthermal corona discharge in liquid around electrodes with ultrasharp tips and around nanowires," Yury Gogotsi informs Nanowerk. "We have demonstrated that plasmas created with 50 nm probe tips or carbon nanotubes – what we have termed nanoscale corona discharge probes or NCDPs – dispersed in solutions allow simultaneous chemical analysis of multiple dissolved elements within nanoseconds. Time-resolved optical emission spectroscopy of NCDPs demonstrates narrow spectral lines that prove very useful for simple yet sensitive multi elemental analysis, thus opening new possibilities in chemical detection, environmental monitoring, medicine, and many other applications." | |
 |
|
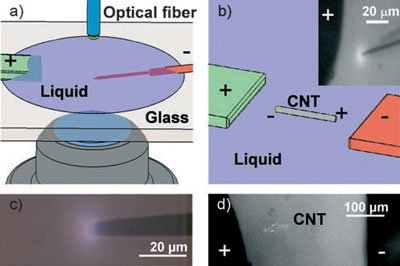
| Direct (a) and remote (b) setups for micro- and nanoscale corona discharge OES. a) An electrically wired probe with tip sharpened to 100 nm diameter immersed in a grounded fluid. Discharge is observed by microscope and spectra are collected through an optical fiber. b) Disconnected high aspect ratio, highly polarizable object, such as a carbon nanotube, in fluid. Bias on remote electrodes induces fields and creates corona discharge. Inset shows an induced corona around a 4 mm carbon microfiber in water. c,d) Optical micrographs of typical coronas around a wire tip and a bundle of multiwalled carbon nanotubes. (Reprinted with permission from Wiley) | |
|
Gogotsi, a professor in the Department of Materials Science and Engineering, heads the A.J. Drexel Nanotechnology Institute at Drexel University, together with colleagues Prof. Gary Friedman from Drexel's Department of Electrical and Computer Engineering, and Dr. Alexander Gutsol (currently with Chevron), Prof. Alexander Fridman (director of the A.J. Drexel Plasma Institute), and first author, PhD student, David Staack |
|
| Gogotsi says that the optical emission spectroscopy (OES) method proposed by the Drexel scientists can be applied for ultrafast time resolved multi elemental analysis of liquid in microfluidic reactors, living biological systems, or environmental sensors and for diagnostics of femtoliter volumes with one micrometer or better spatial resolution. "Using this method, we have detected part-per-million concentrations of sodium, calcium, and other elements in aqueous solutions." | |
| In comparison to LIBS, the researchers found that the observed spectra are 1) of better quality 2) have significantly smaller analytical volumes and 3) are accomplished using drastically simpler, smaller and less expensive equipment and materials. Furthermore, OES can be performed remotely, using nanorods and nanotubes dispersed in fluid. | |
| In their paper, the team describes how in a typical experiment, a tungsten wire with a tip sharpened to below 50 nm radius was used to generate the corona discharge (alternatively, carbon fibers with a diameter of approximately 4 µm, quartz pipettes with 0.5?1 µm tips covered by a gold film, and template-grown noncatalytic chemical vapor deposition carbon nanotubes of 200 nm diameter were employed). Negative corona discharges at the tips have been demonstrated in all cases. The pulsed voltage source provides 2?30 kV pulses 10?500 ns in duration at approximately 30 Hz repetition rate, achieving negative corona for a 50 nm radius tip with as little as 3 kV. | |
 |
|
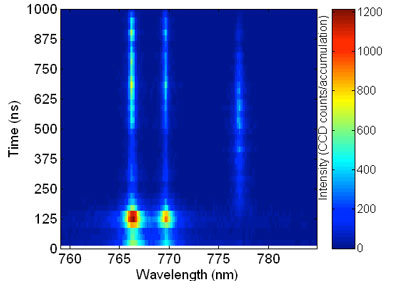
| Time resolved spectra with 25 ns resolution of 10mM KCl aqueous solution. Spectral range shows simultaneous acquisition of three peaks: two due to K (766.5, 769.9) and a single peak due to O (triplet around 777.4). (Reprinted with permission from Wiley) | |
| "Considering existing theories of discharge initiation in liquids and our current experiments, there are several factors that contribute to the reasons why the NCDP is different from the streamer coronas previously observed in liquid and specifically why the two initial stages of the negative corona are observed" Gogotsi explains. "Our study is the first which simultaneously combines 1) short rise-time voltages, 2) nanosecond-duration pulses, 3) high temporal resolution emission spectra, and most importantly 4) nanoscale tips." | |
| He then points out that the nanoscale tip allows for a slower temporal evolution of the discharge in several ways: "First, there are no pre-existing microbubbles on the surface that facilitate breakdown. Second, although the power density remains constant as the electrode size is reduced to the nanoscale, relative losses increase with increasing surface-to-volume ratio S/V≈1/r, analogous to the size stabilization we have observed for microscale plasma discharges in air. Lastly, with nanoscale electrodes, field emission processes are more controllable, and there is no need to rely on unknown asperities or preexisting bubbles as in macroscale discharge devices. As such, the discharge can be operated with only very small over voltages (voltages in excess of the field emission threshold)." | |
| The Drexel team is hopeful that the OES nanoscale probes may open a new era in micro/nanoscale chemical, environmental and biological sensing and detection techniques. | |
| "Just like the discovery of atomic force microscopy changed the world of microscopy, the OES nanoscale probes may change the world of chemical analysis, replacing large and expensive instruments, which are used for elemental analysis or measurements of cation concentration in thousands of labs worldwide, with simple, portable and very inexpensive tools that also add analytical capabilities not available today, e.g., fast simultaneous quantitative analysis of multiple cations in solution," says Gogotsi. | |
| With regards to the non-thermal plasma in liquid, nanoscale corona discharge OES is presented only as the first of many potential applications for this newly discovered tool; applications in nanopatterning and surface functionalization as well as tools for cellular surgery are readily conceivable. Gogotsi and his colleagues expect this research to affect a broad spectrum of fields ranging from pharmaceuticals and biomedicine to nanotechnology and fundamental plasma chemistry. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
