| Posted: Sep 26, 2008 | |
Sensor paints herald new era of responsive materials |
|
| (Nanowerk Spotlight) 'Smart' is the key buzz word used by materials engineers when they describe the future of coatings, textiles, building structures, vehicles and just any material that you can think of. Materials are made 'smart' when they are engineered to have properties that change in a controlled manner under the influence of external stimuli such as mechanical stress, temperature, humidity, electric charge, magnetic fields etc. Smart materials have some form of sensor capability that detects a change in the material or its environment that then triggers some kind of action. The European project Inteltex for instance is developing a new, multifunctional textile that could be used as a wallpaper to detect temperature changes or chemical leakage or that could be used in medical and protective wear to monitor body temperature and mechanical stress. | |
| Not only for use in smart materials but as general sensor materials, especially for monitoring large areas, the development of materials that act as 'chemical paints' – or coatings – by responding to a (bio)chemical parameter with a change in their optical properties has developed into an exciting new field. In a typical application, the object of interest is painted and the color or fluorescence of the paint is monitored by methods of optical imaging. This technique represents a simple but exciting new technology to monitor (bio)chemical and even physical parameters over relatively large areas and in real time without having to look at only a minute sample through a microscope. | |
| Writing a review in Advanced Materials ("Sensor Paints"), Dr. Otto S. Wolfbeis, a professor at the University of Regensburg in Germany focuses on current applications of sensor coatings, the state of the research and the challenges that are faced for robust sensor coatings. | |
| Adopting a characterization technique commonly used in chemical imaging, sensor paints work by detecting fluorescent emissions. Wolfbeis describes the basic workings of this technique: "The term 'paint' refers to a material that consists of a solvent, a polymeric support (sometimes referred to as a binder), and an indicator dye. This cocktail can be painted or sprayed onto the surface of the object of interest or can be manufactured as a thin film that, after solvent evaporation, is placed on the system to be studied. The indicator dye in the paint undergoes diffusional equilibration with its environment, and the analyte of interest thereby modulates the fluorescence of the paint, which is then photographed or imaged." | |
| Intrinsically fluorescent chemical species, such as reduced nicotinamide adenine dinucleotide (NADH), flavines, chlorophylls, and the like, can be visualized directly by fluorescent imaging – fluorescent field imaging for instance is used to scan large areas of ocean for polycyclic aromatic hydrocarbons, chlorophylls, and algae. | |
| There are, however, numerous analytes that neither have an intrinsic luminescence nor can be rendered luminescent with the help of labels or probes. Such parameters include oxygen, pH, carbon dioxide, ammonia, and glucose. It is highly desirable, though, to study the distribution of the concentration of such species over larger areas, for example, on human skin, in an organ to be transplanted, the mat on a sea floor, or the local distribution of oxygen in a bioreactor. In this case, the system to be analyzed can be painted with a sensor substance that responds to the respective parameter of interest. | |
| In his review article, Wolfbeis lists five specific areas with examples of sensor paint applications: | |
| Marine research | |
| pH-sensitive paints have been applied to monitor the pH of seawater and its gradients. The use of respective sensor paints enabled to create images of the gradients of oxygen and pH. One study ("Fluorescent Imaging of pH with Optical Sensors Using Time Domain Dual Lifetime Referencing") for instance has shown that pH can be imaged using fluorescent sensor paints, and this was used to visualize pH and oxygen in marine sediments. The paint was deposited on the wall of a kind of aquarium that was filled with sediment and seawater. | |
| High-throughput screening and combinatorial microbiology | |
| Sensor paints for pH and for oxygen have been deposited on the bottom of wells of microtiter plates in which bacteria were cultivated. The growth of bacteria (an important parameter in high-throughput screening for antibiotics) in a set of wells can be imaged even in millisecond intervals by imaging the fluorescence of the single spots in each well. The effects were also shown to be useful for highly parallel screening of enzyme activities, to monitor respirometric activity, and in toxicological assays based on inhibition of enzymatic activity. | |
| Medicine and biology | |
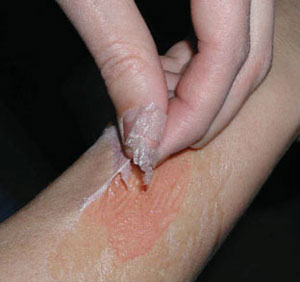
| Sensor paints can be used to study the oxygenation of (tumorous) skin and to monitor the beneficial effect of photodynamic therapy of skin cancer. A biocompatible paint containing the (orange-colored) oxygen probe is sprayed onto skin and then covered with an oxygen-impermeable material (so to prevent access of ambient oxygen). A thin film is formed after evaporation of the solvents. Thereafter, the temperature of the skin is raised to >40°C so as to facilitate rapid equilibration of oxygen between skin and sensor paint. | |
|
|
| Oxygen-sensitive paint for imaging the distribution of oxygen on skin. (Reprinted with permission from Wiley) | |
| Another use of oxygen-sensitive materials is in the food industry where they could be used in packaging to indicate spoiled food (see: Intelligent inks – now you see them, now you don't). | |
| Progress has also been made to sense physiologically important species such as glucose. In this case, the enzyme glucose oxidase in the paint triggers the oxidation of glucose and causes the formation of hydrogen peroxide which is then visualized with a hydrogen peroxide-sensitive paint. | |
| Pressure-sensitive paints | |
| Wolfbeis writes that a most impressive and widespread application consists of the use of oxygen-sensitive paints to measure air pressure. The technique is based on the fact that the partial pressure of oxygen reflects the total pressure of air as long as its fraction in air remains constant. Hence, optical sensor layers can be used as pressure-sensitive paints. | |
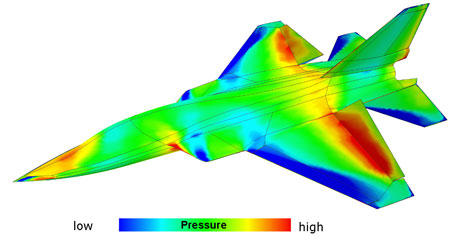
| Research on air pressure-sensitive paints (PSP) has originated with various aerodynamic research facilities all over the world. A pressure image can be obtained by photographing the luminescence after photoexciting the paint with an appropriate light source, allowing for real-time monitoring of air pressures of models in a wind tunnel, for example. The paints are so thin and smooth that they do not disturb the air flow around the model, making them vastly superior to traditional mechanical sensors. | |
| While conventional pressure measurement methods are only capable to measure the pressure distribution on discrete points on the surface by installing pressure sensors such as taps and transducers, the two-dimensional PSP method provides a measurement of the entire surface. In addition, due to a better spatial resolution, disturbances in flow are observable. A major drawback of conventional pressure measurement technique is avoided by using PSP: The location of pressure taps must be determined prior to model manufacturing, at a time when the flow pattern is not known exactly. Also there are restrictions to drill holes in thin wind tunnel models in order to install taps and transducers. These restrictions do not apply for a PSP coating applied to the surface of the model with a spray gun. | |
|
|
| Distribution of surface pressure on the model of an aircraft covered with a pressure-sensitive paint. Blue areas indicate high pressure, red areas indicate low pressure. (Source: German Aerospace Center (DLR), Institute of Aerodynamics and Flow Technology) | |
| Temperature-sensitive paints | |
| Temperature-sensitive paints represent an attractive alternative to infrared-based thermo-imaging. As in the case of pressure-sensitive paints, the indicator dye is incorporated into a polymer binder and the paint is sprayed onto the surface to be imaged. Then, the solvent is evaporated and luminescence imaged following photoexcitation from light sources such as xenon lamps or batteries of LEDs. | |
| Scientists are already working on developing multiple sensing coatings. Wolfbeis writes that materials have already been reported that can sense both pH and oxygen, or temperature and oxygen, or carbon dioxide and oxygen. Imaging of oxygen and temperature simultaneously is particularly interesting since this also allows pressure sensors to be intrinsically compensated for the effect of temperature. Moreover, dual sensors reduce the need for large sample volumes (such as blood). | |
| Although the vast potential of these novel coating materials is clear, there are still numerous challenges that researchers need to overcome before sensor paints move out of the lab and see large scale applications in a real-world setting. The limitations and problems that Wolfbeis lists include photolability, poor adhesion to surfaces, the use of toxic solvents which creates recycling problems, a lack of tolerance of high temperatures (above 200°C), low pressure sensitivity, unsatisfactory response times (they are expected to lie below 0.01 seconds). And finally, it would be attractive to have probes that respond to gases other than oxygen, for example, nitrogen – but none have been discovered yet. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com.
