| Posted: Mar 29, 2012 | |
Graphene nanosensor tattoo on teeth monitors bacteria in your mouth |
|
| (Nanowerk Spotlight) Detection of very small amounts of a chemical contaminant, virus or bacteria in food systems is an important potential application of nanotechnology. The exciting possibility of combining biology and nanoscale technology into sensors holds the potential of increased sensitivity and therefore a significantly reduced response-time to sense potential problems (see our previous Nanowerk Spotlight: "Nanotechnology and microbiology"). | |
| Bacterial contamination in general, is a major problem affecting both developing and developed countries and the rapid and sensitive detection of pathogenic bacteria at the point of care is extremely important; especially given that more and more pathogens are becoming immune to antibiotics (DARPA even seeks to replace antibiotics with rapidly adaptable nanotherapeutics). | |
| Early detection of pathogenic bacteria is critical to prevent disease outbreaks and preserve public health. This has led to urgent demands to develop highly efficient strategies for isolating and detecting this microorganism in connection to food safety, medical diagnostics, water quality, and counter-terrorism. | |
| Limitations of most of the conventional diagnostic methods are the lack of ultrasensitivity and the delays in getting results. One problem is that many infectious agents have very low minimum infective doses, requiring detection mechanisms that offer extremely high sensitivities. Another problem is the time delay between taking samples and getting the results: Current methods for the detection of pathogenic contaminants involve the collection and pre-processing of analyte samples – biological specimen, food samples etc. – and then performing laboratory-based assays. It will be extremely advantageous to have sensing systems that can be directly integrated with the sources of contamination or points of infection to provide an in situ monitoring/detection bacterial presence. | |
| A team of scientists, led by Fiorenzo Omenetto at Tufts University and Michael C. McAlpine at Princeton University, have now developed a novel approach to interfacing passive, wireless graphene nanosensors onto biomaterials via silk bioresorption. They have reported their finding in the March 27, 2012 online edition of Nature Communications ("Graphene-based wireless bacteria detection on tooth enamel"). | |
 |
|
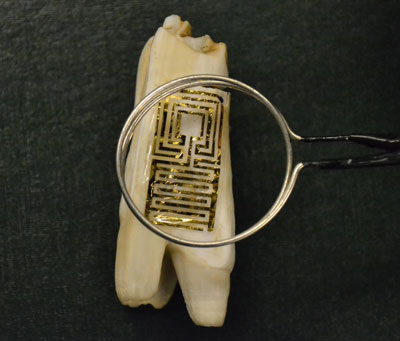
| Optical image of the graphene wireless sensor biotransferred onto the surface of a tooth. (Image: McAlpine Group, Princeton University) | |
| "Graphene is capable of highly sensitive analyte detection," says Manu S. Mannoor, a graduate student in McAlpine's group and the paper's first author. "In our paper, we demonstrate that graphene can be printed onto water-soluble silk. This in turn permits intimate biotransfer and direct interfacing of graphene nanosensors with a variety of substrates including biological tissues and hospital IV bags to provide in situ monitoring and detection of bacterial contamination and infection." | |
| The nanoscale nature of graphene allows for high adhesive conformality after biotransfer and highly sensitive detection. The team demonstrates their nanosensor by attaching it to a tooth for battery-free, remote monitoring of respiration and bacteria detection in saliva. | |
| Such an approach of direct interfacing of nanosensors onto biomaterials could revolutionize areas ranging from health quality monitoring to adaptive threat detection. | |
| Mannoor explains the fabrication technique to Nanowerk: "First, we printed graphene nanosensors onto water-soluble silk thin-film substrates. The graphene is then contacted by interdigitated electrodes, which are simultaneously patterned with an inductive coil antenna. Finally, the graphene/electrode/silk hybrid structure is transferred to biomaterials such as tooth enamel or tissue, followed by functionalization with bifunctional graphene?AMP biorecognition moeities." | |
| Silk thin-films serve as an ideal 'temporary tattoo' platform owing to their optical transparency, mechanical robustness, biotransferability, flexibility and biocompatibility. Omenetto's research group has been conducting research with silk composite materials for biosensing applications for a while now and has already reported a promising path towards the development of a new class of metamaterial-inspired implantable biosensors and biodetectors (see "Implantable silk metamaterials could advance biomedicine, biosensing"). | |
| The resulting device architecture is capable of extremely sensitive chemical and biological sensing, with detection limits down to a single bacterium, while also wirelessly achieving remote powering and readout. Potential applications for this kind of 'biotransferrable' sensors could include on-body health quality monitoring, hospital sanitation monitoring, and food safety analysis and may provide a first line of defence against pathogenic threats at the point of contamination. | |
| "What we were able to demonstrate is only a prototype, 'first generation' platform that served as a proof of concept for the in situ bacterial contamination monitoring by direct interfacing of graphene nanosensors with a variety of substrates including biological tissues," says Mannoor. "Future challenges include mainly improving the selectivity of the detection system to be able to distinguish between various species of pathogenic bacteria. Reducing the sensor form factor is also another challenge facing the future development of the sensor." | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
