| Posted: Nov 05, 2008 | |
The gripping potential of DNA nanotechnology |
|
| (Nanowerk Spotlight) DNA, the fundamental building block of our genetic makeup, has become an intense nanotechnology research field. DNA molecules can serve as precisely controllable and programmable scaffolds for organizing functional nanomaterials in the design, fabrication, and characterization of nanometer scale electronic devices and sensors. The reason why DNA could be useful in DNA nanotechnology for the design of electric circuits is the fact that it actually is the best nanowire in existence – it self-assembles, it self-replicates and it can adopt various states and conformations (see: DNA electronics in nanotechnology or The long road to molecular electronics could be paved with DNA). Then of course there is a rich body of work on DNA use in nanorobotics such as Ned Seeman's work at NYU (see: Nanorobotic arm to operate within DNA sequence). | |
| The most basic and simplest form of DNA mechanical devices that are expected to be the first to demonstrate some close-to-reality functions are DNA tweezers. This concept was first introduced in 2000 by scientists at Bell Labs and Oxford University ("A DNA-fuelled molecular machine made of DNA"). To keep this type of tweezers running, two fuel DNA strands are alternately added to a buffered solution that contains the tweezers. These fuels are basically two stretches of complementary DNA, one of which closes the tweezers and the other opens them. | |
| The exciting potential applications for DNA tweezers include their use in constructing various molecular devices dedicated to repairing a functional unit in a cell, harnessing the delivery of drug molecules to pathogenic cells, or assembling nanoscale devices. | |
| There have been several earlier versions of similar DNA devices that can be operated to have open and close actions but it still has been a challenge to make them behave like real tweezers that can be handled to grasp and transfer an object. A team of Chinese scientists has now demonstrated a very simple design to fabricate a close-to-reality 'grasp' and 'release' function for a pair of DNA tweezers. | |
| "The most challenging part to make this tweezer function available has to do with the structural simplicity of DNA tweezers that only have two mechanical arms – which is not a good setup for stably capturing something" Dr. Zhaoxiang Deng tells Nanowerk. "We have successfully circumvented this challenge thanks to the helical wrapping action of the DNA target around the tweezers’ arms during the formation of Hoogsteen bonds ("Reprogramming DNA-Directed Reactions on the Basis of a DNA Conformational Change"), which greatly increases the probability for the target to be held between the tweezers' arms." | |
| Based on this, Deng, a professor in the Department of Chemistry at the University of Science & Technology of China in Hefei, Anhui Province, together with his group, has successfully built a pair of DNA tweezers that can capture, hold, and release an object with easy control. They have reported their findings in the October 14, 2008 online edition of Journal of American Chemical Society ("Catch and Release: DNA Tweezers that Can Capture, Hold, and Release an Object under Control"). | |
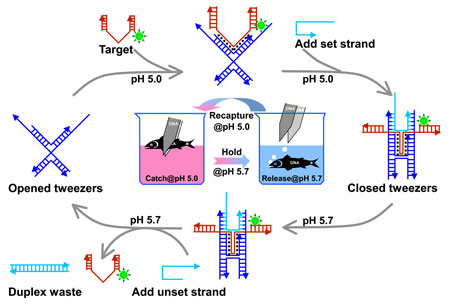 |
|
| A target (in red), which is a special DNA assembly, binds to opened tweezers (in blue) at pH 5.0 due to a special interaction called Hoogsteen hydrogen bonding formed between the target and the two arms of the tweezers. A set strand (in cyan) is then added to zip the tweezers close so that the target will be tightly held between the two arms of tweezers at an increased pH of 5.7. An unset DNA strand (in cyan) is then introduced, which has a sequence fully complementary to the set strand, to rip away the closing set strand from the tweezers by a process called toehold (a single stranded overhang on the set strand after closing the tweezers) mediated strand displacement and open the arms wide so that the target can be released. The whole process can go round and round with the duplex formed between the set and unset strands as one of the wastes (the other waste is water as a result of acid-base reactions during pH switchings). The target is labeled with a fluorescent tag (in green) so that this catch and release process can be easily monitored by a fluorescent gel imaging technique. Note that the helical shape of the target strand wrapping around the tweezers’ arms is not reflected in this drawing for simplicity, but we have to stress here that it plays a key role for the target to be stably captured. (Image: Dr. Deng) | |
| The team re-engineered the two arms of previously reported DNA tweezers ("Addressable Molecular Tweezers for DNA-Templated Coupling Reactions") to take advantage of the formation of a DNA triplex via Hoogsteen hydrogen bonding between a target DNA and the two arms. Deng explains the three steps that are involved to operate such a device: | |
| "Firstly, four DNA strands are annealed to form opened tweezers. A DNA target pre-assembled from three DNA strands is introduced. The single stranded part of this target has a polypyrimidine sequence that will form triplexes with the tweezers’ arms at pH 5.0. Secondly, a DNA set (locker) strand is then employed to close the tweezers by hybridizing with two single strand overhangs on the tweezers’ arms. The target has five consecutive adenines in the middle to ensure sufficient flexibility when the tweezers are closed. The pH is then switched to 5.7, and at this increased pH the tweezers still firmly hold the target via a closing action and some remaining Hoogsteen bonding. Finally, an unset (unlocker) strand that fully complements the set strand is added to remove the set strand from the closed tweezers. This process utilizes some yet unpaired bases on the set strand to initialize a strand displacement reaction. The tweezers then are opened, and the DNA target is released." | |
| The dimension of the newly designed tweezers in the 'closed' state was about 2 x 4 x 10 cubic nanometers, with each arm being about 3.7 nm long corresponding to a helical turn in a B-form DNA duplex, a common format of double stranded DNA found in our body. Most importantly, the new tweezers assembled by Deng's team could now be easily handled to capture a special DNA target and hold it and release it at will. | |
| As Deng explains, no sophisticated facilities are required to operate this tiny device. "All you need is two extra fuel DNA strands that played exactly the same roles as those used for the millennium version of the DNA tweezers by Yurke et al. One difference is that our tweezers need the two fuel strands to be in phase with pH switchings that basically control the 'stickiness' of the target to the tweezers' arms in responding to a 'hold' or 'release' command." | |
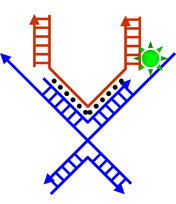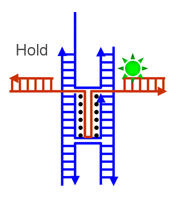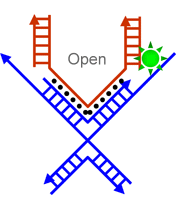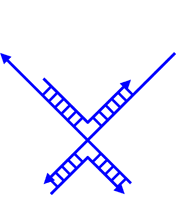 |
|
| Schematic illustration of the operation of a pair of DNA tweezers (Image: Dr. Deng) | |
| The scientists caution that, although they have successfully demonstrated the operations of their tweezer, it is not a design that is ready for real-world applications yet. "For example, the current design of our tweezers requires a relatively narrow pH switching range from 5.0 to 5.7" says Deng. "Although this was not a problem for a proof-of-concept demonstration, a wider pH range, and therefore less pH control requirements, will make the tweezers easier to operate." | |
| He also points out that the tweezers currently can not hold all the target molecules at the optimized pH of 5.7. This necessitates further work towards obtaining an even sharper transition between the hold and release states. "With technological advances in related areas, we have all reasons to believe the next generation of our tweezers will be able to function even better" says Deng. | |
| He points out that the DNA target can also be thought of as a carrier so that other objects that do not have similar interactions with the tweezers can still be handled after being chemically loaded (conjugated) on the target. "Therefore, the applications of our work are not limited to DNA based objects. We will also try to immobilize the tweezers on micro/nanosized spheres (such as magnetic beads) so that they can be easily recovered from operation buffers with the help of a magnet when their running efficiency gets lower due to the accumulation of waste materials in the buffer." | |
| Deng mentions other applications, where the tweezers may act as a relay between two special processes whose mutual communications can be turned on or off by holding or releasing any signaling substances – chemical or physical objects – transmitted between them (such as what happens around a nerve terminal). "Also, if you imagine a nanofactory, you would need a mechanical hand to deal with many different tasks such as taking one part from an upstream worker, processing it and then handing it over to a downstream worker. In this process, a device like the one we have invented might find some uses even though it might be too early to think about these questions." | |
| The team is currently considering the integration of different DNA mechanical devices including the tweezers toward multiplexed and cooperative functions. Deng mentions that the simple demonstration devices that currently exist can only perform a simple task at a time; something which is not suitable for the construction of highly advanced nanofabrication systems and other nano devices. He notes that the ability to be integrated into larger assemblies is an inherent merit of DNA structures, which makes it possible to combine different functional units in a single platform and also allows them to be individually addressable after being coded using DNA sequences. | |
| According to Deng, the work that is being performed his laboratory on building functional DNA nanodevices is very early days: "A lot of fundamental work still has to be carried out before we will see any interesting real-world applications." | |
| He says that to use future DNA nanomachines, or even nanorobots, in a complex setup will require highly sophisticated forms of control mechanisms that integrate and synchronize vastly disparate elements and functions: "The most difficult thing is that all these devices can have dramatically different structural features, require different operations and generate totally different forms of outputs. Here, we are not talking about how to simply put individual parts together, which is relatively easy to do if they are all made of DNA, but how to make them work in a cooperative manner so that one device’s action can be a control over another and vice versa." | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com.
