Showing Spotlights 137 - 144 of 240 in category All (newest first):
 Graphene with its distinctive band structure and unique physiochemical properties - such as exceptionally low intrinsic electrical resistivity, high surface area, rapid electrode kinetics and good mechanical properties - is considered an attractive material for analytical electrochemistry. However, one of the key technical challenges for the use of graphene as functional material in device applications is the integration of nanoscale graphene onto micro- or millimeter sized sensing platforms. With a new methodology, a team from Florida International University was able to integrate graphene onto three-dimensional (3D) carbon microstructure arrays with good uniformity and controllable morphology.
Graphene with its distinctive band structure and unique physiochemical properties - such as exceptionally low intrinsic electrical resistivity, high surface area, rapid electrode kinetics and good mechanical properties - is considered an attractive material for analytical electrochemistry. However, one of the key technical challenges for the use of graphene as functional material in device applications is the integration of nanoscale graphene onto micro- or millimeter sized sensing platforms. With a new methodology, a team from Florida International University was able to integrate graphene onto three-dimensional (3D) carbon microstructure arrays with good uniformity and controllable morphology.
Apr 24th, 2012
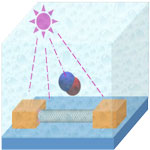 Single-walled carbon nanotubes arguably are the ultimate biosensor among nanoscale semiconducting materials due to their high surface-to-volume ratio and unique electronic structure. After more than a decade of excitement though, more and more researchers in the nanotube field believe that pristine SWCNTs are very limited as a sensing material. Ironically the ultrahigh sensitivity of SWCNTs is easily compromised by various unintentional contaminants from the device fabrication process as well as the ambient environment. As a result, significant efforts have been focused on all kinds of ways to functionalize or decorate nanotubes with other species in order to improve their sensitivity. Researchers have now shown that applying continuous in situ ultraviolet light illumination during gas detection can enhance a SWCNT-sensor's performance by orders of magnitude under otherwise identical sensing conditions.
Single-walled carbon nanotubes arguably are the ultimate biosensor among nanoscale semiconducting materials due to their high surface-to-volume ratio and unique electronic structure. After more than a decade of excitement though, more and more researchers in the nanotube field believe that pristine SWCNTs are very limited as a sensing material. Ironically the ultrahigh sensitivity of SWCNTs is easily compromised by various unintentional contaminants from the device fabrication process as well as the ambient environment. As a result, significant efforts have been focused on all kinds of ways to functionalize or decorate nanotubes with other species in order to improve their sensitivity. Researchers have now shown that applying continuous in situ ultraviolet light illumination during gas detection can enhance a SWCNT-sensor's performance by orders of magnitude under otherwise identical sensing conditions.
Apr 10th, 2012
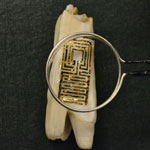 Early detection of pathogenic bacteria is critical to prevent disease outbreaks and preserve public health. This has led to urgent demands to develop highly efficient strategies for isolating and detecting this microorganism in connection to food safety, medical diagnostics, water quality, and counter-terrorism. A team of scientists has now developed a novel approach to interfacing passive, wireless graphene nanosensors onto biomaterials via silk bioresorption. The nanoscale nature of graphene allows for high adhesive conformality after biotransfer and highly sensitive detection. The team demonstrates their nanosensor by attaching it to a tooth for battery-free, remote monitoring of respiration and bacteria detection in saliva.
Early detection of pathogenic bacteria is critical to prevent disease outbreaks and preserve public health. This has led to urgent demands to develop highly efficient strategies for isolating and detecting this microorganism in connection to food safety, medical diagnostics, water quality, and counter-terrorism. A team of scientists has now developed a novel approach to interfacing passive, wireless graphene nanosensors onto biomaterials via silk bioresorption. The nanoscale nature of graphene allows for high adhesive conformality after biotransfer and highly sensitive detection. The team demonstrates their nanosensor by attaching it to a tooth for battery-free, remote monitoring of respiration and bacteria detection in saliva.
Mar 29th, 2012
 Optical fibers have revolutionized telecommunications by providing higher performance, more reliable telecommunication links with ever decreasing bandwidth cost. In parallel with these developments, fiber-optic sensor technology has been a major user of technologies associated with the optoelectronic and fiber optic communications industry. Today, with the rapid advance of communications and especially sensing applications, there is an ever increasing need for advanced performance and additional functionalities. This, however, is difficult to achieve without addressing fundamental fabrication issues related to the integration onto optical fibers of advanced functional materials at the micro- and nanoscale. Solving these technical problems will open up the possibility of developing multifunctional labs integrated onto a single optical fiber, exchanging information and combining sensorial data. This could result in auto diagnostic features as well as new photonic and electro-optic functionalities useful in many strategic sectors such as optical processing, environment, life science, safety and security.
Optical fibers have revolutionized telecommunications by providing higher performance, more reliable telecommunication links with ever decreasing bandwidth cost. In parallel with these developments, fiber-optic sensor technology has been a major user of technologies associated with the optoelectronic and fiber optic communications industry. Today, with the rapid advance of communications and especially sensing applications, there is an ever increasing need for advanced performance and additional functionalities. This, however, is difficult to achieve without addressing fundamental fabrication issues related to the integration onto optical fibers of advanced functional materials at the micro- and nanoscale. Solving these technical problems will open up the possibility of developing multifunctional labs integrated onto a single optical fiber, exchanging information and combining sensorial data. This could result in auto diagnostic features as well as new photonic and electro-optic functionalities useful in many strategic sectors such as optical processing, environment, life science, safety and security.
Mar 27th, 2012
 Various types of nanostructures are used in the development of nanosensors: nanoparticles, nanotubes, nanorods, two-dimensional materials like graphene, embedded nanostructures, porous silicon, and self-assembled materials. For instance, gas sensors often operate by detecting the subtle changes that deposited gas molecules make in the way electricity moves through a surface layer. Researchers have fabricated gas sensors based on carbon nanotube-based field effect transistors, which can detect electrical potential changes around them. While these and related sensing schemes can be all-electronic - i.e., not requiring optical readout - they all require sophisticated nanolithographic techniques to isolate, identify, and integrate electrical contact to the active nanosensor. Researchers have now presented a nanoscale 3D architecture that can afford highly sensitive, room temperature, rapid response, and all-electronic chemical detection.
Various types of nanostructures are used in the development of nanosensors: nanoparticles, nanotubes, nanorods, two-dimensional materials like graphene, embedded nanostructures, porous silicon, and self-assembled materials. For instance, gas sensors often operate by detecting the subtle changes that deposited gas molecules make in the way electricity moves through a surface layer. Researchers have fabricated gas sensors based on carbon nanotube-based field effect transistors, which can detect electrical potential changes around them. While these and related sensing schemes can be all-electronic - i.e., not requiring optical readout - they all require sophisticated nanolithographic techniques to isolate, identify, and integrate electrical contact to the active nanosensor. Researchers have now presented a nanoscale 3D architecture that can afford highly sensitive, room temperature, rapid response, and all-electronic chemical detection.
Mar 21st, 2012
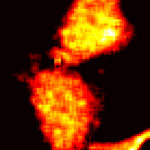 Green Fluorescent Protein (GFP) - originally found in a jellyfish - has played a crucial role in life science research, providing insights to many fundamental questions that have paved the way to the biology and medicine of the future. Since the mid-1990s, when the protein was successfully cloned, GFP can be found in research laboratories worldwide used as a visual marker of gene expression and protein localization, easily observed via light (optical) microscopy. GFP can be linked to other proteins and is primarily used to track dynamic changes in living cells. In 2008, biologists who discovered and developed the protein as a laboratory tool won a Nobel Prize for their work. Researchers in Spain have now demonstrated how GFP can also act as an efficient nano-thermometer inside cells.
Green Fluorescent Protein (GFP) - originally found in a jellyfish - has played a crucial role in life science research, providing insights to many fundamental questions that have paved the way to the biology and medicine of the future. Since the mid-1990s, when the protein was successfully cloned, GFP can be found in research laboratories worldwide used as a visual marker of gene expression and protein localization, easily observed via light (optical) microscopy. GFP can be linked to other proteins and is primarily used to track dynamic changes in living cells. In 2008, biologists who discovered and developed the protein as a laboratory tool won a Nobel Prize for their work. Researchers in Spain have now demonstrated how GFP can also act as an efficient nano-thermometer inside cells.
Mar 19th, 2012
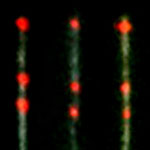 Knowing the distribution of DNA binding proteins along the genome is very informative and can tell scientists about the state of gene expression at the time of measurement. These DNA-binding proteins include transcription factors which modulate the process of transcription, various polymerases, nucleases which cleave DNA molecules, and histones which are involved in chromosome packaging in the cell nucleus. Previously, researchers demonstrated the viability of a single-molecule approach to directly visualize and map protein binding sites on DNA using fluorescent quantum dots, allowing multicolor, nanometer-resolution localization. Now, they have shown that proteins bound to DNA can be located very accurately by direct imaging. The precision of these measurement presents new opportunities for contextual genomic research on the single-molecule level.
Knowing the distribution of DNA binding proteins along the genome is very informative and can tell scientists about the state of gene expression at the time of measurement. These DNA-binding proteins include transcription factors which modulate the process of transcription, various polymerases, nucleases which cleave DNA molecules, and histones which are involved in chromosome packaging in the cell nucleus. Previously, researchers demonstrated the viability of a single-molecule approach to directly visualize and map protein binding sites on DNA using fluorescent quantum dots, allowing multicolor, nanometer-resolution localization. Now, they have shown that proteins bound to DNA can be located very accurately by direct imaging. The precision of these measurement presents new opportunities for contextual genomic research on the single-molecule level.
Mar 12th, 2012
 The Venus flytrap (Dionaea muscipula) is a carnivorous plant that catches and digests little insects. Its trapping mechanism consists of a series of tiny hairs at the crease where the plant's two leaves join. When a fly or spider walk across these hairs, touching two or more of them in succession, the two leaves will close quickly enough - within hundreds of milliseconds - to prevent its escape. Now, researchers have used it as inspiration for a new biomimetic robot made with artificial muscles. The device offers promise in the development of electrically stimulated artificial muscle that could be implanted in people to help overcome muscular disease or paralysis.
The Venus flytrap (Dionaea muscipula) is a carnivorous plant that catches and digests little insects. Its trapping mechanism consists of a series of tiny hairs at the crease where the plant's two leaves join. When a fly or spider walk across these hairs, touching two or more of them in succession, the two leaves will close quickly enough - within hundreds of milliseconds - to prevent its escape. Now, researchers have used it as inspiration for a new biomimetic robot made with artificial muscles. The device offers promise in the development of electrically stimulated artificial muscle that could be implanted in people to help overcome muscular disease or paralysis.
Mar 6th, 2012
 Graphene with its distinctive band structure and unique physiochemical properties - such as exceptionally low intrinsic electrical resistivity, high surface area, rapid electrode kinetics and good mechanical properties - is considered an attractive material for analytical electrochemistry. However, one of the key technical challenges for the use of graphene as functional material in device applications is the integration of nanoscale graphene onto micro- or millimeter sized sensing platforms. With a new methodology, a team from Florida International University was able to integrate graphene onto three-dimensional (3D) carbon microstructure arrays with good uniformity and controllable morphology.
Graphene with its distinctive band structure and unique physiochemical properties - such as exceptionally low intrinsic electrical resistivity, high surface area, rapid electrode kinetics and good mechanical properties - is considered an attractive material for analytical electrochemistry. However, one of the key technical challenges for the use of graphene as functional material in device applications is the integration of nanoscale graphene onto micro- or millimeter sized sensing platforms. With a new methodology, a team from Florida International University was able to integrate graphene onto three-dimensional (3D) carbon microstructure arrays with good uniformity and controllable morphology.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





