| May 19, 2020 | |
Injecting biomedical electronics for monitoring and therapy of body organs |
|
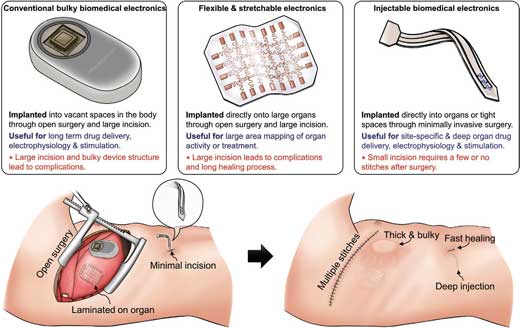
| (Nanowerk Spotlight) Implantable electronic devices range from sensors, gastric and cardiac pacemakers, cardioverter defibrillators, to deep brain, nerve, and bone stimulators. These devices are interfaced with the human body to extract precise medical data and to interfere with tissue function by providing electrical stimuli. Long-term implants present specific engineering challenges, including low energy consumption and stable performance. Furthermore, most electronic materials have poor bio- and cytocompatibility, resulting in immune reactions and infections. | |
| Initially, electronics which can interface with the body typical were planar, stiff and bulky silicon wafer-based devices and not well suited to interface with the soft, curvilinear, and dynamic environment that biology presents. To overcome these problems, one of the device classes that researchers have fabricated are relatively novel, miniaturized needle-like carriers in high aspect ratio structures. They are used to deliver tiny sensors and stimulation tools inside the body via minimally invasive injection or insertion into a specific area of an organ. | |
 |
|
| Comparison of state-of-the-art implantable electronics in different forms. (Reprinted with permission by Wiley-VCH Verlag) (click on image to enlarge) | |
| A recent review in Advanced Materials ("Injectable Biomedical Devices for Sensing and Stimulating Internal Body Organs") summarizes the latest developments in materials, designs, and manufacturing techniques in the field of injectable biomedical devices, highlighting unique applications and demonstrations of viable clinical tools that were applied in various internal organs. | |
| Injectable biomedical devices is a rapidly developing field driven by advancements in semiconductor technology, allowing wafer-based electronics and optoelectronics that are fabricated at micro- and nanoscales to be transformed into high aspect ratio forms. | |
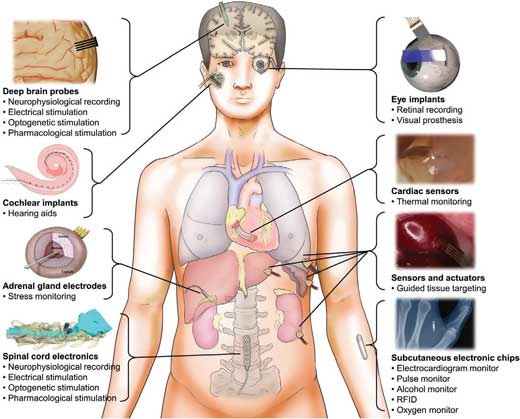
| By now, various classes of biomedical devices in high aspect ratio forms have been applied to almost all body parts. For example, deep brain stimulation uses metal or silicon electrodes shaped into high aspect ratio, injectable format to target neurons in the deep regions of the brain with minimal damage to other brain regions. Ultrathin needle-like eye implants replace the damaged retina lying inside the eyeball with minimal surgical opening which would otherwise create leakages of the vitreous fluid. | |
 |
|
| Diagram of the human anatomy and different classes of injectable biomedical devices for sensing and stimulating internal body organs. Deep brain probes provide recording and stimulating capabilities of the brain with minimal damage to neuronal tissues. The insertable electrodes of the cochlear implants translate external sounds to electrical signals for hearing aid. Polymeric probes injected into the adrenal glands measure cortisol concentration levels for stress monitoring. Soft electronics are inserted into the spinal cords to measure neuronal activity and stimulate neurons. Visual prosthesis is achieved using flexible optoelectronic system injected through the eye. Injectable temperature sensors accurately detect lesions along the heart walls. Piezoelectric devices placed on injectable probes provide tissue mechanics for cancer targeting. Subcutaneous injectable devices can record, track and stimulate body information in safe and reliable manner. (Reprinted with permission by Wiley-VCH Verlag) (click on image to enlarge) | |
| Microelectromechanical systems (MEMS) can also be implemented into these wafer-based injectable biomedical devices to create complex 3D geometries, such as fluidic channels and optical waveguides. More recently, the ability to fabricate such advanced devices on polymeric substrates created a low-cost and thinner form of needles for mechanically compliant operation inside the body. Simultaneous operation of different sensor types as well as simulation tools could be realized via transfer printing various devices onto the polymeric needle tip. | |
| For optical stimulations, fiber optics have dominated deep tissue stimulations. Recent developments in fiber techniques embedded electronic tools other than optical waveguide into the fiber, such as electrodes and microfluidic channels, all within the microscale fiber diameter. Efforts to further reduce device sizes down to the nanoscale could be realized using syringe-injectable mesh electronics. | |
| Nanowires and nanomesh forms of metal electrodes and semiconductors could be injected deep into the body parts using conventional syringe needles as their physical size is extremely small to fit inside the syringe needle tips. | |
| Integrating multiple electronic and optoelectronic functionalities within microscale needle-like architecture demands understanding of both conventional microfabrication processes and relatively new fabrication methods, such as transfer printing, thermal drawing, and polymer molding techniques. | |
| Advances in multimaterial structure fabrication technologies have resulted in increasing number of multifunctional, high aspect ratio biomedical tools in the past decade. In this review, the authors describe the recent advancements of these injectable classes of biomedical devices applied for each body part, reviewing the strategies used to achieve these goals and different classes of materials and electronic devices developed for various applications, specifically: | |
|
|
|
 |
|
| Injectable electronics integrated with optoelectronics and microfluidics for the brain. Left: A multichannel neural probe with embedded microfluidic channels for simultaneous in vivo neural recording and drug delivery. SEM image of the fabricated neural probe showing the tip of a shank with eight iridium microelectrodes and a microfluidic outlet. Middle: A silicon 2D optrode array capable of delivering light to multiple sites using SU-8 as a waveguide core. SEM image of the fabricated optrodes showing the tip of a single shank with the optical waveguide core and microelectrode array. Right: Monolithically integrated µ-LEDs on silicon neural probes. Image of an implantation-ready silicon neural probe with recording electrodes and integrated µ-LEDs (inset, scale bar, 15 µm). (Reprinted with permission by Wiley-VCH Verlag) (click on image to enlarge) | |
| Concluding their review, the authors caution that many of the research results reviewed by them are preliminary viability testing of new classes of electronics with high aspect ratio designs. Further research and development in optimizing the designs and features should prepare the reviewed injectable biomedical tools for human trials. | |
| They also summarize the possible concerns and suggested solutions related to the development of injectable biomedical electronics and stress that more work is needed to conduct the safety assessments associated with long term use of these devices before they can be ready for clinical use. | |
| A remaining challenge of injectable biomedical electronics is the elimination of wires for obtaining data or providing power. To date, the practical uses of novel biomedical electronics have been delayed by the lack of wireless communication capabilities, which are especially difficult as they target organs deep inside the body. Commercially successful devices, such as cochlear implants and insertable cardiac monitors, all include wireless capabilities; however, these devices include rigid electronic chips that need to be encased in relatively bulky designs. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
