| Posted: Feb 13, 2014 | |
Nanotechnology needle arrays for drug delivery |
|
| (Nanowerk Spotlight) It is estimated that as much as 99% of medicinal molecules administered during a therapy don't reach their targets and subsequently stay in the body of the patient. Some of these molecules can be very toxic, especially cancer drugs, and the potential side effects of many therapeutic drugs can be downright frightening – just read the instruction leaflet that comes with your pills. | |
| These effects often occur when a drug is active throughout the body, not just where and when it is needed. As opposed to having patients simply swallow a pill, health care professionals have long envisioned delivering specific quantities of medicines to targeted areas of the body, thereby increasing the treatment’s effectiveness while reducing side effects. In order to achieve this, a 'vehicle' of sorts is needed to safely and accurately deliver the medicine to the desired location within the body. | |
Nanotechnology for drug delivery |
|
| The ultimate goal of nanotechnology-enabled drug delivery, especially with regard to cancer therapy, is to ferry most of the administered drug to the target, while eliminating the accumulation of the drug at any non-target tissues. | |
| Nanomedicine applications with targeted nanoparticles are expected to revolutionize cancer therapy. The use of such nanoparticles to deliver therapeutic agents is currently being studied as a promising method by which drugs can be effectively targeted to specific cells in the body, such as tumor cells. | |
| Biological barriers – the skin, mucosal membranes, the blood-brain barrier and cell/nuclear membranes – seriously limit the delivery of drugs into the desired sites within the body, resulting in a low delivery efficacy, poor therapeutic efficacy, and high cost. | |
| Nanomedicine researchers have developed numerous biological, chemical, and physical strategies to overcome these barriers. A new review paper in Advanced Materials highlights recent advanced physical approaches for transdermal and intracellular delivery ("Advanced Materials and Nanotechnology for Drug Delivery"). | |
Transdermal delivery |
|
| The core idea of microneedle patches is to achieve improved vaccine efficiency and effectiveness – over the needle and syringe – through precise targeting of vaccines to skin strata, achieved by device miniaturization utilizing micro- and nanotechnologies (read more: "Needle-free, painless vaccinations with nanopatches"). | |
| At the early stage of the development, microneedle arrays were made of silicon and metals. These microneedles are often a few hundred micrometers in length and with very sharp tips to ensure reliable penetration into the epidermal and upper dermal layers of the skin for drug delivery. With these solid microneedles, drugs are often dry-coated on their surface for subsequent delivery to the skin. | |
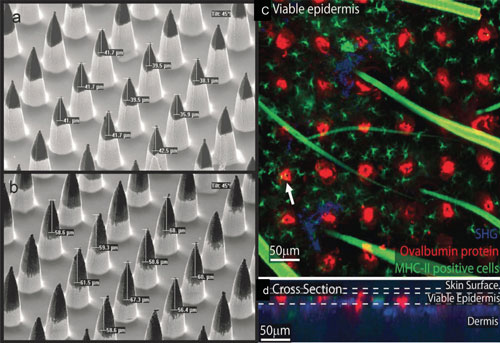 |
|
| a,b) SEM images of silicon microneedles coated with ovalbumin (OVA) protein. The dark signal on the tips of the microneedles indicate dry-coated DNA vaccine, which can be dissolved and released in the epidermal and dermal layers once the microprojections are inserted into the skin. c,d) Fluorescence microscopy images of the release of Cy5-OVA (red) from the microneedle patch applied to murine skin. The image shows a 0.176 µm 2 region of the patched area consisting of 36 projections sites. MHC-II positive cells were stained using an FITC (green) stain. The second harmonic generation of collagen is also shown (blue). d) A cross-sectional view of six coating delivery sites. Colocalization of MHCII positive cells and delivered vaccine is shown (the arrow head in Figure c). ). (Reprinted with permission from Wiley-VCH Verlag GmbH & Co. KGaA) (click image to enlarge) | |
| Although positive results have been achieved with these silicon and metal microneedle arrays for transdermal delivery, there are some safety and cost concerns, including: 1) the fabrication of these microneedles often requires expensive materials and production technologies such as deep reactive ion etching (DRIE) and laser cutting; and 2) the possible breakage of the small microneedles inside the skin leads to safety problems. | |
| To overcome these disadvantages, one approach is to choose biocompatible and biodegradable polymers as the materials for manufacturing microneedle arrays. | |
| Although dissolvable microneedles possess many advantages and have been demonstrated to work effectively, these dissolvable polymers generally have relatively weak mechanical properties and researchers are working to improve their mechanical strength to ensure reliable, consistent, and reproducible penetration into the human skin for future practical applications. | |
Intracellular delivery |
|
| Delivery of biomolecules – e.g., cell probes, genes, peptides, drugs, and proteins – into living cells is of great importance for gene and cell therapy. These therapies are promising for curing many diseases which are otherwise difficult to be treated (see for instance: "Nanotechnology based stem cell therapies for damaged heart muscles"). | |
| It is also often necessary to deliver fluorescent probes into cells for studying biological processes at the molecular level inside living cells. However, these molecules have a low capability of penetrating cell membranes because of a variety of reasons including large size, surface charge, instability, hydrophilicity, etc. | |
| "Inspired by the success of employing microneedle arrays for drug delivery, we first reported using a nanoneedle array to actively and mechanically disrupt cell membranes for intracellular delivery," says Xianfeng Chen, an assistant professor at the City University of Hong Kong and lead author of the review. "Transdermal and intracellular delivery has one common point. For transdermal delivery, the outmost layer of skin, stratum corneum limits the diffusion of most materials, molecules, genes and drugs to the skin. For intracellular delivery, the cell membrane plays a similar role, therefore, as microneedles can be used to pierce the stratum corneum to achieve transdermal delivery, one should be able to use a similar approach for intracellular delivery." | |
| However, if microneedles are applied to cells, because of their large geometry, the cells might be killed easily. Therefore, nanoneedles – nanowires or carbon nanotubes – need to be designed for intracellular delivery. | |
| Chen and his co-authors conclude that the approach of applying nanoneedle arrays for intracellular delivery is currently still at its starting stage. More systematic studies are required to demonstrate the advantages of the technology. In addition, it is essential to carry out research to optimize the manufacturing of these nanoneedle arrays and design parameters for optimal and reproducible results. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
