Showing Spotlights 25 - 32 of 110 in category All (newest first):
 The surface force balance (SFB) provides measurements of surface and colloidal forces in liquids such as electrostatic surface forces, van der Waals forces, and solvation forces. Until now, the SFB required mica sheets as the substrate for measurements. This was the only material available in an atomically smooth state over centimeter-scale areas as well as being optically transparent as required for the optical interferometry. By replacing the mica sheets with graphene, electrically conducting and atomically smooth surfaces for the measurement of surface forces have now been created.
The surface force balance (SFB) provides measurements of surface and colloidal forces in liquids such as electrostatic surface forces, van der Waals forces, and solvation forces. Until now, the SFB required mica sheets as the substrate for measurements. This was the only material available in an atomically smooth state over centimeter-scale areas as well as being optically transparent as required for the optical interferometry. By replacing the mica sheets with graphene, electrically conducting and atomically smooth surfaces for the measurement of surface forces have now been created.
Dec 10th, 2014
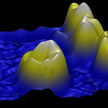 Researchers have demonstrated a new imaging technique that is a marriage between two powerful methods and it promises simultaneous spatial and elemental information of the samples down to the atomic scale. By combining scanning tunneling microscopy (STM) with synchrotron X-ray microscopy, there is now an instrument (SX-STM) that has the potential to perform all the applications of STM and X-rays in a single setting at the ultimate atomic limit.
Researchers have demonstrated a new imaging technique that is a marriage between two powerful methods and it promises simultaneous spatial and elemental information of the samples down to the atomic scale. By combining scanning tunneling microscopy (STM) with synchrotron X-ray microscopy, there is now an instrument (SX-STM) that has the potential to perform all the applications of STM and X-rays in a single setting at the ultimate atomic limit.
Oct 27th, 2014
 The desire to identify materials and their properties to understand complex systems and better engineer their functions has been driving scanning probe microscopies since their inception. Both atomic force microscopy (AFM) and Raman spectroscopy are techniques used to gather information about the surface properties and chemical information of a sample. There are many reasons to combine these two technologies, and this application note discusses both the complementary information gained from the techniques and how a researcher having access to a combined system can benefit from the additional information available.
The desire to identify materials and their properties to understand complex systems and better engineer their functions has been driving scanning probe microscopies since their inception. Both atomic force microscopy (AFM) and Raman spectroscopy are techniques used to gather information about the surface properties and chemical information of a sample. There are many reasons to combine these two technologies, and this application note discusses both the complementary information gained from the techniques and how a researcher having access to a combined system can benefit from the additional information available.
Mar 6th, 2014
 Surface metrology and characterization is ever more critical for overall product performance in wide ranging applications across the semi-conductor, LED, data storage, medical and automotive industries. 3D optical microscopes are among the fastest and most accurate imaging systems on the market today, and are employed in these industries for rapid and precise process monitoring, product development, and research. However, there are instances where they have performance limitations and the benefits of scanning probe/atomic force microscopy provide a clear advantage.
Surface metrology and characterization is ever more critical for overall product performance in wide ranging applications across the semi-conductor, LED, data storage, medical and automotive industries. 3D optical microscopes are among the fastest and most accurate imaging systems on the market today, and are employed in these industries for rapid and precise process monitoring, product development, and research. However, there are instances where they have performance limitations and the benefits of scanning probe/atomic force microscopy provide a clear advantage.
Feb 6th, 2014
 Measuring and mapping mechanical properties of live cells is of high importance in today's biological research. raditionally, force spectroscopy and force volume are the most commonly used modes to quantitatively measure mechanical forces at the nanometer scale. Unfortunately, both techniques have suffered from slow acquisition speed and a lack of automated tools to analyze the hundreds to thousands of curves required for good statistics. This application note reviews recent progress in mapping the properties of soft samples such as cells and gels with force volume and PeakForce QNM and the use of the newest NanoScope and NanoScope Analysis features to collect and analyze the data from these techniques.
Measuring and mapping mechanical properties of live cells is of high importance in today's biological research. raditionally, force spectroscopy and force volume are the most commonly used modes to quantitatively measure mechanical forces at the nanometer scale. Unfortunately, both techniques have suffered from slow acquisition speed and a lack of automated tools to analyze the hundreds to thousands of curves required for good statistics. This application note reviews recent progress in mapping the properties of soft samples such as cells and gels with force volume and PeakForce QNM and the use of the newest NanoScope and NanoScope Analysis features to collect and analyze the data from these techniques.
Dec 30th, 2013
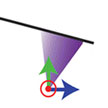 The realization of a three-dimensional atomic force microscopy portends exciting research directions across nanoscience and nanotechnology. Demonstrations to date have been limited by the indirect means that are required to extract a three-dimensional force vector from the traditional 1D observable in AFM (i.e., cantilever deflection). Existing 3D AFM techniques require recording thousands of frequency shift curves at different lateral locations followed by off-line integration (to yield energy) and lateral differentiation (to yield lateral force). This procedure is inherently slow. In new work, researchers now report 3D force measurements based on a 3D local observable, rather than on cantilever deflection alone.
The realization of a three-dimensional atomic force microscopy portends exciting research directions across nanoscience and nanotechnology. Demonstrations to date have been limited by the indirect means that are required to extract a three-dimensional force vector from the traditional 1D observable in AFM (i.e., cantilever deflection). Existing 3D AFM techniques require recording thousands of frequency shift curves at different lateral locations followed by off-line integration (to yield energy) and lateral differentiation (to yield lateral force). This procedure is inherently slow. In new work, researchers now report 3D force measurements based on a 3D local observable, rather than on cantilever deflection alone.
Oct 14th, 2013
 Optical imaging of nanoscale objects, whether it is based on scattering or fluorescence, is a challenging task due to reduced detection signal-to-noise-ratio and contrast at sub-wavelength dimensions. While advances in light microscopy have led to techniques that can image individual nanoparticles, these methods rely on relatively sophisticated and expensive microscopy systems. Researchers have now created a field-portable fluorescence microscopy platform installed on a smartphone for imaging of individual nanoparticles as well as viruses using a light-weight and compact opto-mechanical attachment to the existing camera module of the cellphone.
Optical imaging of nanoscale objects, whether it is based on scattering or fluorescence, is a challenging task due to reduced detection signal-to-noise-ratio and contrast at sub-wavelength dimensions. While advances in light microscopy have led to techniques that can image individual nanoparticles, these methods rely on relatively sophisticated and expensive microscopy systems. Researchers have now created a field-portable fluorescence microscopy platform installed on a smartphone for imaging of individual nanoparticles as well as viruses using a light-weight and compact opto-mechanical attachment to the existing camera module of the cellphone.
Sep 17th, 2013
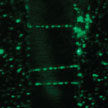 Direct visualization and manipulation of individual carbon nanotubes (CNTs) in ambient conditions is of great significance for their characterizations and applications. However, the direct visualization, location, and manipulation of individual CNTs is extremely difficult due to their nanoscale diameters. The observation of individual CNTs usually requires electron microscopes under high vacuum. Researchers now have proposed a facile way to realize optical visualization of individual carbon nanotubes and, based on that, macroscale manipulation of individual carbon nanotubes that could be carried out under an optical microscope.
Direct visualization and manipulation of individual carbon nanotubes (CNTs) in ambient conditions is of great significance for their characterizations and applications. However, the direct visualization, location, and manipulation of individual CNTs is extremely difficult due to their nanoscale diameters. The observation of individual CNTs usually requires electron microscopes under high vacuum. Researchers now have proposed a facile way to realize optical visualization of individual carbon nanotubes and, based on that, macroscale manipulation of individual carbon nanotubes that could be carried out under an optical microscope.
Jul 22nd, 2013
 The surface force balance (SFB) provides measurements of surface and colloidal forces in liquids such as electrostatic surface forces, van der Waals forces, and solvation forces. Until now, the SFB required mica sheets as the substrate for measurements. This was the only material available in an atomically smooth state over centimeter-scale areas as well as being optically transparent as required for the optical interferometry. By replacing the mica sheets with graphene, electrically conducting and atomically smooth surfaces for the measurement of surface forces have now been created.
The surface force balance (SFB) provides measurements of surface and colloidal forces in liquids such as electrostatic surface forces, van der Waals forces, and solvation forces. Until now, the SFB required mica sheets as the substrate for measurements. This was the only material available in an atomically smooth state over centimeter-scale areas as well as being optically transparent as required for the optical interferometry. By replacing the mica sheets with graphene, electrically conducting and atomically smooth surfaces for the measurement of surface forces have now been created.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





