| Posted: Sep 16, 2010 | |
Pinning neurons in place with nanopillars |
|
| (Nanowerk Spotlight) Studies of the interface between nanostructures and live cells have been increasing rapidly in the past few years. Because the size of nanostructures is often comparable to internal organelles inside the cell, those nanostructures are very useful in serving as sensors to detect biological events inside a live cell (see for instance: "Live cell imaging with biodegradable quantum dot nanocomposites"). Even more futuristic concepts envision the incorporation of functional nanodevices inside living cells (see: "Future bio-nanotechnology will use computer chips inside living cells"). | |
| For neuroscientists, monitoring the electrical signaling within neural networks is a fundamental issue. It has proven to be very challenging to monitoring individual neuron activities in a neuronal network for an extended time – weeks or months – which demands stable and specific neuron-electrode correspondence. Unfortunately for the scientists, neurons tend to migrate as far as hundreds of micrometers, often moving outside the range of the electrode that is supposed to monitor it. | |
| "Electric signals generated by a neuron can be detected extracellularly if there is an electrode in close contact" Bianxiao Cui, an assistant professor in the Department of Chemistry at Stanford University, explains to Nanowerk. "However, it has been difficult to consistently measure the activity of the same neuron over a long-term period. This difficulty is partly due to neuron mobility and partly due to lack of neuron-to-electrode specificity. As a result, patterned electrodes or transistors are not always monitoring the activity of the same neuron as neurons move around." | |
| While previous techniques try to prevent neuron migration by chemically or physically confining them, Cui and her collaborators seek to engineer unique nanostructures that foster, rather than impose, residence of neuron cell bodies atop the electrode of interest. | |
| In their recent work, reported in the September 3, 2010 online issue of Nano Letters ("Noninvasive Neuron Pinning with Nanopillar Arrays"), the researchers describe the use of nanopillar arrays to pin the position of neurons in a noninvasive manner. This work provides a new means to anchor a neuron to an external electrode for long-term electric measurement. | |
| Cui's team found that vertical nanopillars non-invasively inhibit the migration of neuron cells that are growing attached to them. Neurons in close contact with the nanopillars show significantly reduced mobility and are essentially pinned to the nanopillars. | |
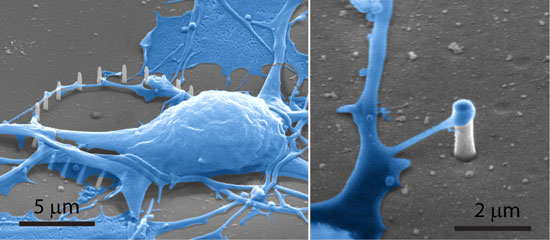 |
|
| SEM of cells cultured on Si and SiO2 nanopillar substrates. Left: An SEM image shows a SiO2 pillar engulfed by the cell membrane. Right: An SEM image shows a protruding of a neuron reaching a Si nanopillar. (Image: Cui Lab, Stanford University) | |
| "Despite this pinning effect, neurons growing on nanopillars show similar growth patterns to those seen on a flat substrate" says Cui. "Within the parameter regime that we have tested – 75-400 nm in diameter and 700 nm to 2 µm in height – cell survival rate and the pinning effect do not seem to depend on the size of the pillars." | |
| She notes that, if patterned on top of microelectrodes, vertical nanopillars would serve as neuron traps for long-term neuronal network study with multielectrode arrays and also improve the neuron-to-electrode contact. | |
| While previous studies had used larger structures (tens of micrometers) to act as physical barriers preventing neurons escaping from a cage or a fence, the nanopillars fabricated by the Stanford team are very small with dimensions comparable to some cellular organelles (∼150 nm diameter and ∼1 µm height). | |
| "Neurons actively engulf the nanopillars and prefer anchoring to nanopillars over the flat surface, and therefore, the nanopillars inhibit the cell migration by acting as 'attractants'" explains Cui. | |
| The researchers fabricated their nanopillars by ion-beam or e-beam induced platinum deposition using a dual-beam focused ion beam (FIB)/scanning electron microscope (SEM) system. They chose platinum material for its biocompatibility and potential to directly measure the electrical activities of attached neurons. | |
| "We fabricated the nanopillar patterns on top of the electrodes of a customized multielectrode array substrate (MEA) in order to anchor neurons at the electrode locations" explains Cui. "Since non-transparent MEA electrodes make it difficult to observe nanopillars or neurons on top of them using optical microscopy, we also fabricate nanopillars in the transparent area of the fused quartz substrate to demonstrate the cell pinning effect of the nanopillars." | |
| Besides the specific problem of establishing stable and specific neuron-to-electrode correspondence, the research team also envisions two important potential applications. | |
| "The first potential application is axon guidance using patterned substrate" says Cui. "Because vertical nanopillars serve as attractants for neuron cell bodies and their axons, the growing axon can be guided by the patterned nanopillars on the substrate into desired geometries. The second potential application is to use the vertical nanopillars, which are made of platinum material, to serve as tiny electrodes to record or stimulate the neuron that are attached to them." | |
| Given the tiny size of the sensors, the signal strength and the intrinsic noise of the biological environment will be a big challenge in designing functional sensors for practical use. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
