Showing Spotlights 57 - 64 of 559 in category All (newest first):
 Nanotechnology has the ability to completely transform the health care sector, particularly in developing countries like South Africa, where access to effective healthcare is still a challenge for millions of people living in poverty-stricken environments. Many African countries, despite having policies and strategies in place, struggle to allocate sufficient resources for research in nanomedicine. Most of the research conducted on the subject in Africa is focused on academic interests, rather than practical applications.
Nanotechnology has the ability to completely transform the health care sector, particularly in developing countries like South Africa, where access to effective healthcare is still a challenge for millions of people living in poverty-stricken environments. Many African countries, despite having policies and strategies in place, struggle to allocate sufficient resources for research in nanomedicine. Most of the research conducted on the subject in Africa is focused on academic interests, rather than practical applications.
Jan 15th, 2023
 Its significance of the protein corona for biomedical applications lies in its role of imparting a unique biological identity to the nanoparticle, which could be very different from the pristine nanoparticle surface. Robust characterization of the identity and abundance of the protein corona is entirely dependent on liquid chromatography coupled to mass spectroscopy. Unfortunately, the variability of this technique for the purpose of protein corona characterization remains poorly understood.
Its significance of the protein corona for biomedical applications lies in its role of imparting a unique biological identity to the nanoparticle, which could be very different from the pristine nanoparticle surface. Robust characterization of the identity and abundance of the protein corona is entirely dependent on liquid chromatography coupled to mass spectroscopy. Unfortunately, the variability of this technique for the purpose of protein corona characterization remains poorly understood.
Nov 3rd, 2022
 Therapeutic nucleic acids (TNAs) and nucleic acid nanoparticles (NANPs) are designed to enact specific intracellular responses beneficial for various biomedical applications. TNAs become especially attractive to researchers and clinicians since their functional versatility, programmability and modularity show great promise for treating disorders such as viral infections, cancers, and genetic disorders. Scientists explore how the immune responses to NANPs change with various factors such as dimensionality (3D vs. 2D), composition (DNA vs. RNA), and functionalization with TNAs.
Therapeutic nucleic acids (TNAs) and nucleic acid nanoparticles (NANPs) are designed to enact specific intracellular responses beneficial for various biomedical applications. TNAs become especially attractive to researchers and clinicians since their functional versatility, programmability and modularity show great promise for treating disorders such as viral infections, cancers, and genetic disorders. Scientists explore how the immune responses to NANPs change with various factors such as dimensionality (3D vs. 2D), composition (DNA vs. RNA), and functionalization with TNAs.
Aug 15th, 2022
 The defenses of the body's immune system tend to destroy synthetic nanoparticles and frequently they are captured and removed from the body within few minutes. This, of course, is a major barrier to the use of nanotechnology in medicine. In order to evade the host immune response, researchers are demonstrating that the combination of nanomaterials and biomimetic strategies can change diverse nanomaterials from 'distinguishable state' to 'camouflage state' and thereby improving technologies for nanomedicine applications.
The defenses of the body's immune system tend to destroy synthetic nanoparticles and frequently they are captured and removed from the body within few minutes. This, of course, is a major barrier to the use of nanotechnology in medicine. In order to evade the host immune response, researchers are demonstrating that the combination of nanomaterials and biomimetic strategies can change diverse nanomaterials from 'distinguishable state' to 'camouflage state' and thereby improving technologies for nanomedicine applications.
Aug 9th, 2022
 Researchers have developed an optothermal platform with five manipulation modes - printing, tweezing, rotating, rolling, and shooting - using a single Gaussian laser beam. Switching between the different modes is achieved by simply tuning the optical power or the distance between laser and object, and can be automated through software programming. This novel multimodal manipulation technique relies on the coordination of multiple thermal forces instead of light-matter interactions, i.e., the mechanism of traditional optical tweezers.
Researchers have developed an optothermal platform with five manipulation modes - printing, tweezing, rotating, rolling, and shooting - using a single Gaussian laser beam. Switching between the different modes is achieved by simply tuning the optical power or the distance between laser and object, and can be automated through software programming. This novel multimodal manipulation technique relies on the coordination of multiple thermal forces instead of light-matter interactions, i.e., the mechanism of traditional optical tweezers.
Jul 25th, 2022
 Inspired by living biological systems, which can sense, process information, and actuate in response to changing environmental conditions, researchers are exploring engineered living hydrogels. The convergence of engineering, biology, and materials science over the past 20 years is providing unprecedented opportunities to integrate living cells into these hydrogels. This integration yields engineered living hydrogels with the capabilities of self-replication, self-regulation, and environmental responsiveness.
Inspired by living biological systems, which can sense, process information, and actuate in response to changing environmental conditions, researchers are exploring engineered living hydrogels. The convergence of engineering, biology, and materials science over the past 20 years is providing unprecedented opportunities to integrate living cells into these hydrogels. This integration yields engineered living hydrogels with the capabilities of self-replication, self-regulation, and environmental responsiveness.
Jul 11th, 2022
 Spontaneous charging of interfaces between water and hydrophobic media is a mysterious feature whose nature and origin - despite many efforts to explain them - are still not fully understood. Many spectroscopic results and interpretations along with computational work are not consistent with one another and a consensus on the nature and origin of interfacial charging has not been reached. A clear understanding of this puzzling phenomenon needs a variety of approaches from diverse perspectives, which, hopefully will all add up and be pieced together towards a complete description of the full picture.
Spontaneous charging of interfaces between water and hydrophobic media is a mysterious feature whose nature and origin - despite many efforts to explain them - are still not fully understood. Many spectroscopic results and interpretations along with computational work are not consistent with one another and a consensus on the nature and origin of interfacial charging has not been reached. A clear understanding of this puzzling phenomenon needs a variety of approaches from diverse perspectives, which, hopefully will all add up and be pieced together towards a complete description of the full picture.
Jul 7th, 2022
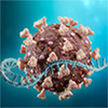 The success of mRNA technology in the development of a COVID-19 vaccine has become a groundbreaking development in the fight against cancer, as well as in the development of new vaccines against many other infectious diseases, including HIV. Future nanotechnology-based mRNA vaccines could allow the delivery of multiple antigen-targeted vaccines in one vaccine lipid nanoparticle to protect against multiple diseases, thereby reducing the number of vaccines needed to prevent common vaccine-preventable diseases.
The success of mRNA technology in the development of a COVID-19 vaccine has become a groundbreaking development in the fight against cancer, as well as in the development of new vaccines against many other infectious diseases, including HIV. Future nanotechnology-based mRNA vaccines could allow the delivery of multiple antigen-targeted vaccines in one vaccine lipid nanoparticle to protect against multiple diseases, thereby reducing the number of vaccines needed to prevent common vaccine-preventable diseases.
Jul 5th, 2022
 Nanotechnology has the ability to completely transform the health care sector, particularly in developing countries like South Africa, where access to effective healthcare is still a challenge for millions of people living in poverty-stricken environments. Many African countries, despite having policies and strategies in place, struggle to allocate sufficient resources for research in nanomedicine. Most of the research conducted on the subject in Africa is focused on academic interests, rather than practical applications.
Nanotechnology has the ability to completely transform the health care sector, particularly in developing countries like South Africa, where access to effective healthcare is still a challenge for millions of people living in poverty-stricken environments. Many African countries, despite having policies and strategies in place, struggle to allocate sufficient resources for research in nanomedicine. Most of the research conducted on the subject in Africa is focused on academic interests, rather than practical applications.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





