| Posted: Sep 15, 2010 | |||
Photoconductive Atomic Force Microscopy for understanding nanostructures and device physics of organic solar cells |
|||
| (Nanowerk Spotlight – Application Notes) Plastic solar cells are emerging as alternative energy sources for the future because of their potential for cheap roll-to-roll printing, ease of processing, light-weight and flexibility. However, their current performance is still low for practical applications which partially originate from the poor understanding of device physics and nanoscale morphology of the photoactive layer. Photoconductive atomic force microscopy is a powerful characterization tool to better understand the complex optoelectronic and morphological phenomena of organic solar cells at the nanoscale. All data for this work was obtained using the MFP-3D™ Atomic Force Microscope from Asylum Research. | |||
| Introduction – Inorganic and Organic Photovoltaics | |||
| Conversion of the Sun's usable solar energy reaching the surface of the Earth in a single hour into electricity could meet all global energy needs for an entire year (see: "Solar cells get flexible"). With their potential for low cost, light-weight, flexibility, and ease of processing and installation, plastic solar cells are expected to be the next energy generation technology for the future. | |||
| The first solar cells based on organic semiconductors demonstrated by Tang in 1986 had an efficiency of 1.0% (see: "Two-layer organic photovoltaic cell"). Although research in organic solar cells has only really been active within the past few years, the efficiency has now reached 7.7% (see: "Polymer solar cells with enhanced open-circuit voltage and efficiency"). Scientists have made great efforts on designing new materials to harvest more solar photons and new device architectures to optimize the output power, targeting to achieve an efficiency above 10% in the next few years. Although, there has been much development and progress in this field, the technology is still far from practical applications. One of the main obstacles to achieve the higher efficiency is that device physics and how the photoactive morphology impacts the device performance are not fully understood. | |||
| In conventional inorganic photovoltaics, which consist of two layers of p-doped and n-doped semiconductors, the light absorbing materials directly produce free electrons and holes for electric current generation. Due to high charge carrier mobilities, the photoactive layer in inorganic solar cells can be made in the micrometer range. By contrast, in organic solar cells, the light absorption forms a bound electron-hole pair – a so-called exciton. | |||
| To generate the electric power, the exciton must dissociate into free electrons and holes. To dissociate the exciton, an interface of electron donor and electron acceptor counterparts with the right energy level alignments needs to be utilized. For this reason, the photoactive layer made from a mixture of donor and acceptor molecules has been developed, so-called bulk heterojunction structure (bi-continuous networks of donor and acceptor). Under solar irradiation, the photogenerated carriers travel along donor and acceptor phases toward the anode and cathode electrodes, respectively, where they are collected to generate power. Due to much lower charge carrier mobilities, the photoactive layer in organic solar cells is often less than 200nm thick. | |||
| Nanoscale Morphology | |||
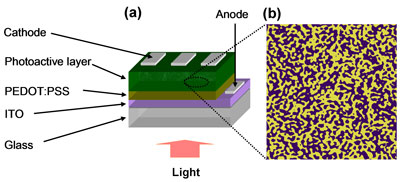
| In organic solar cells, the charge generation and charge transport depend strongly on the nanoscale morphology, defined as the arrangement of the donor and acceptor networks throughout the bulk. At the heart of improving solar cell efficiency is engineering the photoactive morphology to get large interface areas for exciton dissociation and, at the same time, to form continuous donor and acceptor networks for charge transport. Figure 1 provides a schematic drawing of device structure and typical solar cell morphology. The optimal phase of each component should have a domain size between 10nm and 20nm, similar to the exciton diffusion length. | |||
 |
|||
| Figure 1: (a) A typical solar cell structure consisting of a PEDOT:PSS deposited onto a glass/ITO substrate. The photoactive layer is spun cast atop the PEDOT:PSS film, followed by the aluminum cathode electrode evaporation. (b) Topographic image obtained using atomic force microscopy showing phase separation where yellow and purple areas are donor and acceptor phases, respectively. A nanoscale phase separation of donor and acceptor materials in the active layer is needed to achieve efficient exciton dissociation and charge transport. | |||
| To better understand how nanoscale morphology affects charge generation and transport, one needs powerful tools to visualize the phase separation of the two components, as well as to understand the optoelectronic processes occurring in the devices at the nanometer scale. For this purpose, many techniques such as high resolution transmission electron microscopy (TEM) and scanning probe microscopy (SPM) have been deployed. With its wide variety of scanning and measurement modes such as atomic force microscopy (AFM), conductive AFM, transient-resolved electrostatic force microscopy (trEFM), and Kevin probe microscopy (SKPM), SPM can enable simultaneous local probing of morphology, electrical and optoelectronic properties of solar cell materials, establishing a direct correlation of local heterogeneity in nanostructure and photocurrent generation with bulk device performance (see: "Electrical Scanning Probe Microscopy on Active Organic Electronic Devices" and "Heterogeneity in Polymer Solar Cells: Local Morphology and Performance in Organic Photovoltaics Studied with Scanning Probe Microscopy"). | |||
| Photoconductive AFM | |||
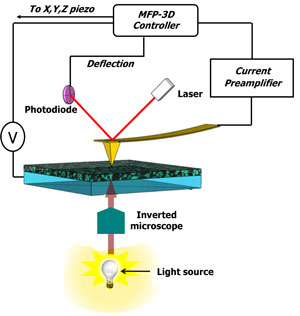
Recently, photoconductive AFM (pcAFM using the MFP-3D AFM from Asylum Research) has been implemented for analyzing solar cell materials (see: "Mapping Local Photocurrents in Polymer/Fullerene Solar Cells with Photoconductive Atomic Force Microscopy" and "Nanostructure and Optoelectronic Characterization of Small Molecule Bulk Heterojunction Solar Cells by Photoconductive Atomic Force Microscopy"). The pcAFM is based on a conductive AFM setup (see: "Nanoscale charge transport and internal structure of bulk heterojunction conjugated polymer/fullerene solar cells by scanning probe microscopy") equipped with a light source (Figure 2). The light is focused on the device through the ITO (indium tin oxide) using an inverted optical microscope and a sample (e.g. a film or device) is loaded in a closed air-tight cell flowed with dry nitrogen. The AFM probe can either sit on a specific point on a sample surface to record the current as a function of an applied bias or the probe can be scanned with a fixed applied bias to provide a current map.
Nanoscale phase separation of donor and acceptor molecules in the photoactive layer is elucidated by imaging electron and hole collection networks at the same location. Detailed description of this research can be found here: "Nanostructure and Optoelectronic Characterization of Small Molecule Bulk Heterojunction Solar Cells by Photoconductive Atomic Force Microscopy". Due to the high work function of the gold-coated silicon probe (~5.1 eV), photogenerated holes are collected by the AFM probe and electrons are collected by the ITO electrode when a bias above open-circuit voltage is applied. This process is reversed when a bias below the open-circuit voltage is applied to the substrate; the photogenerated holes then travel toward the cathode, while the probe tip collects electrons. The applied bias must be small enough so that no charge is injected from the electrodes. Therefore, the photocurrent structure collected at positive and negative biases reveals hole and electron collection networks corresponding to the donor and acceptor phases at the film surface, respectively. An example is shown in Figure 3. The separation of donor and acceptor phases is not apparent in the topography image (Figure 3a). When a bias of +1V is applied to the substrate, the donor domains at the film surface can be visualized at 200nm diameter (Figure 3b). At the same location, the electron collection pathways of 20nm in diameter are imaged at an applied bias of –1V (Figure 3c). Contributing to the low efficiency of the bulk device ("Nanoscale phase separation and high photovoltaic efficiency in solution processed, small molecule bulk heterojunction solar cells"), is the large phase separation of donor and acceptor materials in the blend films which leads to the reduction of interface areas for exciton dissociation and interruption of charge collection pathways. |
|||
 |
|||
| Figure 3: Images of topography (a), current collected at +1V (b) and current collected at –1V (c) of 30:70 DPPBFu:PC71BM films. The photoactive film is deposited on ITO/PEDOT:PSS substrate. The topography and current images were collected using a platinum AFM tip with a bias applied to the substrate. Image sizes are 2µm x 2µm. The data was obtained using the MFP-3D AFM (Asylum Research). | |||
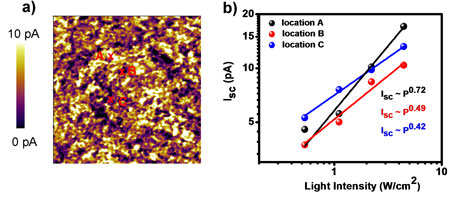
| The pcAFM technique also offers a great opportunity to study nanoscale photophysics using light intensity dependence measurements as described in Figure 4. The heterogeneity in nanostructure and optoelectronics of the photovoltaic materials can be a reason for low performance of bulk devices. Analyzing short-circuit current (Isc) as a function of light intensity sheds light on local variation in the photocurrent generation and recombination. A better understanding of the relationship between nanostructure and optoelectronic properties will help improve the device efficiency ("Nanostructure and Optoelectronic Characterization of Small Molecule Bulk Heterojunction Solar Cells by Photoconductive Atomic Force Microscopy"). The Isc increases with incident light intensity (P), according to a power-law behavior Isc ∼ Pα. The α value is suggestive of the degree of recombination processes. For example, the α in location C is 0.42 as compared with 0.72 in location A, suggesting that the free carrier loss due to charge recombination in location C is much larger than that in location A. Further study, such as external quantum efficiency measurements, can examine the blend composition which results in high photogenerated carrier recombination in C. Comparing this investigation to processing conditions would be the best way for enhancing the efficiency of photovoltaics. | |||
 |
|||
| Figure 4. (a) Short-circuit photocurrent image and (b) light intensity dependence of Isc collected at three locations marked in (a) in DPPBFu:PC71BM films using a gold-coated silicon probe. The data was obtained using the MFP-3D AFM (Asylum Research). | |||
| Spectral Analysis | |||
| A significant advance in the development of pcAFM for organic solar cell characterization is to move from spatial imaging to spectral analysis. Because the photoactive layer is made from a solution of donor and acceptor materials, its morphology is very complex and sensitive to processing conditions. Using the pcAFM technique, complex nanoscale morphology and photocurrent generation can be visualized. | |||
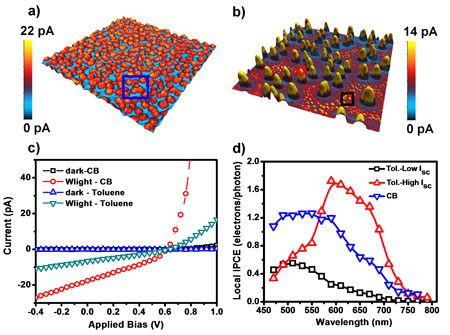
| For example, the photoactive layer of poly[2-methoxy-5-(3,7-dimethyloctyloxy)]-1,4-phenylenevinylene (MDMO-PPV) and [6,6] phenyl-C71-butyric acid methyl ester (PC71BM) spun cast from chlorobenzene solution exhibits less phase separation than that of the films prepared from toluene solution (Figure 5a,b). The smooth surface of chlorobenzene-cast MDMO-PPV:PC71BM films results in more interfaces for exciton dissociation. The average short-circuit photocurrent (Isc) under white light illumination is more efficient than that in toluene-cast films. This result can be confirmed by current-voltage characteristics averaged from different locations as shown in Figure 5c. By using tunable monochromatic lights, we are able to reveal the large phase regions within toluene-cast films (Figure 5b) are PC71BM rich-domains and small particles are distributed by MDMO-PPV polymer. Detailed analysis of external quantum efficiency spectra has elucidated both charge transfer mechanisms existing in the polymer solar cells (Figure 5d); i.e. the holes transfer from the photoexcited acceptor to the donor, and electrons transfer from the photoexcited donor to the acceptor ("Measurement of nanoscale external quantum efficiency of MDMO-PPV:PC71BM solar cells by photoconductive atomic force microscopy"; Appl. Phys. Lett., accepted for publishing.). | |||
 |
|||
| Figure 5. Morphology of MDMO-PPV:PC71BM films spun cast from chlorobenzene (CB) (a) and toluene (Tol) (b). The topography is shown in z-scale of 30nm for (a) and 200nm for (b), while the color is short-circuit photocurrent overlaid on the surface. For photocurrent imaging, the bias is applied to the substrate. Dark and illuminated current-voltage characteristics are recorded as bias applied to the AFM tip (c). Local incident-photon conversion efficiency (IPCE = number of electrons/number of incident photons) of Tol and CB-cast films is shown in (d). Color-coded IPCE spectra in (d) are computed in respective colored boxes in (a) and (b). Image sizes are 10µm x 10µm. The data was obtained using the MFP-3D AFM (Asylum Research). | |||
| Summary | |||
| In summary, we have briefly described the applicability of the pcAFM technique for analyzing solution-processed, polymer and small molecule bulk heterojunction solar cells. Due to the nature of charge generation, transport and collection occurring at the nanometer scale, the useful information on device operation can be lost from macroscopic measurements. | |||
| Currently, pcAFM is the only technique that can provide insights into nanoscale morphology and electrical properties simultaneously and hence elucidate structure-property-performance relationships. However, pcAFM is still in its infancy and requires further development. It is possible to push the limit of the technique to obtain useful information that is relevant to device performance. Together with the complexity of solar cell morphology and photophysics, the quantitative analysis and interpretation of the obtained pcAFM data still remain a challenge. | |||
| By Xuan-Dung Dang and Thuc-Quyen Nguyen. Departments of Chemistry & Biochemistry, Center for Polymers and Organic Solids, University of California, Santa Barbara, CA 93106. E-mails: [email protected], [email protected] | |||
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|||