| Posted: Jul 10, 2007 | |
The challenge of fabricating nanocomposite thin films |
|
| (Nanowerk Spotlight) An incredible amount of research has gone, and still goes, into the understanding of the properties of nanoscale particles. In order to capitalize on that research, scientists and engineers have to take the next step, which is to turn nanoparticle laboratory results into usable materials and devices. One way to use engineered nanoparticles in the real world is in thin films. Nanoparticulate thin films are thin layers, sometimes only a few nanometers thick, of composite materials that contain nanoparticles. These new materials have a wide range of applications such as nanoelectronics, magnetic storage devices, or optical coating. However, most processes used to fabricate thin nanocomposite films with high nanoparticle fillings suffer from random nanoparticle agglomeration causing formation of irregularly shaped nanostructured features within the composite. Unfortunately, the improved mechanical, tribological and electrical properties of the nanocomposites over the host matrix materials (mostly polymers) are only attainable if the nanoparticle inclusions are of uniform size and shape with very good degree of dispersion. A newly developed technique now allows a wide range of control of including metal nanoparticle into polymer matrices in a single step process. | |
| "Recently, nanocomposites of metal nanoparticles in a polymer matrix have received a great deal of interest" >Dr. Abhijit Biswas explains to Nanowerk. "Depending on the metal–polymer composition and structure, such nanocomposites exhibit a unique combination of desirable properties (for example, physical, chemical, mechanical, optical,magnetic and electrical) that are otherwise unattainable. Nanocomposites with metal volume fractions near the percolation threshold are of particular interest. The ability to precisely tailor and optimize the nanocomposite structure near the percolation threshold creates opportunities for addressing a wide range of applications." | |
| Technically speaking, the percolation threshold is the critical fraction of lattice points in a material that must be filled to create a continuous path of nearest neighbors from one side to another. In other words, at the percolation threshold the material becomes highly conductive. As the fraction of the metal in a nanocomposite increases, the nanoparticle separation decreases and at some point reaches this threshold. | |
| "What interests scientists a lot is the region just below the percolation threshold, where the neighboring nanoparticles are still quite densely packed but separated by narrow polymer gaps, the so-called quantum contacts (<5 nm)" says Biswas. "These networks of nanocrystals represent a class of conductors that is neither classically metallic nor a classic dielectric, and offer a host of unique properties relevant to practical applications. These applications include high dielectric constant passives, electromagnetic interference shielding, sensors, detectors or converters designed for a variety of specific purposes with high performance, sensitivity and flexibility." | |
| Biswas, a scientist at the Office of Electronic Miniaturization (OEM) at the University of Alaska Fairbanks, is part of a team that includes Dr. Ilker Bayer (also from the OEM) and Prof. M. Grant Norton from Washington State University at Pullman together with a team of international collaborators from different universities and industry including Motorola. In a recent paper in Nanotechnology (" Networks of ultra-fine Ag nanocrystals in a Teflon AF® matrix by vapor phase e-beam-assisted deposition") this team has reported the ability to fabricate polymer-metal nanocomposite thin films featuring high metal nanoparticle loadings (up to 75 %) with uniform shape and sizes using a vapor phase deposition technique. | |
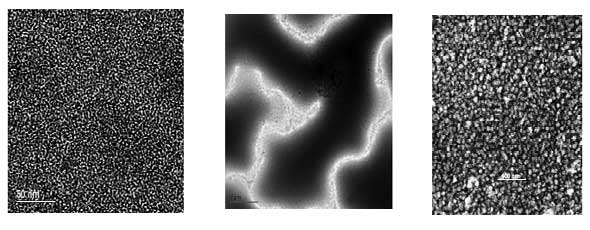 |
Some examples of nanocomposite thin films developed at OEM. Left: Polymer-ceramic nanocomposite for high-value capacitors. Middle: Polymer-polymer nanocomposite for flexible organic electronics. Right: polymer-based Nanocomposite membrane material for water purification. (Images: Dr. Biswas) |
| Biswas says that they have fabricated their nanocomposite thin films using a single step electron-beam-assisted physical vapor deposition. Four different silver nanoparticle volume fillings (20%, 35%, 70% and 75%) were achieved by varying the relative metal–polymer evaporation rates with the formation of highly crystalline silver nanoparticles regardless of the filling ratio. "Our present fabrication technique allows full control over dispersion uniformity of nanoparticles in the polymer network" he says. | |
| "Based on our detailed TEM observations, we strongly believe that the originality of our findings come from the fact that even at very high metal nanoparticle loading fractions, a uniform dispersion of the nanoparticles in the polymer matrix is maintained up to 70% volume filling of nanoparticles" says Biswas. "This is very difficult to attain using other techniques such as sol-gel fabrication." | |
| The inclusion of high volume nanoparticles into polymer matrices using various sol gel and extrusion techniques in a controlled manner is hindered by the fact that these particles tend to agglomerate once a certain filling volume is reached. This causes the formation of irregularly shaped metal clusters with sizes above the nanoscale. | |
| The newly developed technique could be useful for producing a variety of nanostructured materials in the form of special coatings for electromagnetic shielding, anti-microbial coatings (due to silver nanoparticles), embedded capacitor applications in miniature electronic devices and tribological coatings with enhanced elasticity. | |
| "Our method is essentially an e-beam-assisted effusion cell process" says Biswas. "A metal (molybdenum, tantalum or tungsten) crucible bombarded with electrons is used as an effusion cell to evaporate the polymer without decomposing it. The e-beam source can heat the target material to temperatures in excess of 3000°C. Hence, a range of different materials including polymers, metals and ceramic scan be evaporated." | |
| Biswas and his collaborators are pursuing a number of future directions in which applications of nanocomposites will have a considerable impact on various technologies. "Our research can find immediate applications in the fields of energy storage devices, broadband optical absorption for photovoltaics and organic electronics" he says. "Nanocomposites allow the fabrication of otherwise difficult to form structures and their potential for new applications in membrane science and technology for water purification is emerging quickly." | |
| He cautions, however, that in spite of the progress made so far, a number of challenges regarding controlled mixing of different dissimilar components in nanocomposite systems and optimization of the individual properties at the nanoscale still remain to be overcome. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com.
